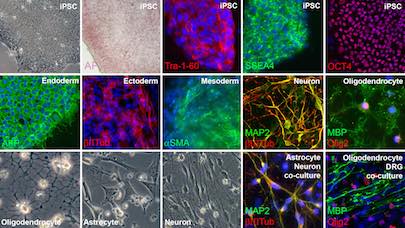
The iPS cell laboratory routinely cultures human embryonic stem cells (hESCs) and human iPS cells in a standardized feeder-free mode (geltrex, Essential E8 medium).
To generate new patient-specific iPS cells from primary cell cultures (e.g. fibroblast), we use a polycistronic lentiviral vector in which codon-optimized reprogramming factors (OCT4, KLF4, SOX2 and cMYC) are co-expressed with the fluorescent marker dTomato under the control of a retroviral promoter (SFFV) which is rapidly silenced in iPS cells. iPSCs are characterized using an enzymatic assay to measure alkaline phosphatase activity, immunocytochemistry and RT-PCR to study expression of the pluripotent genes, like Nanog, Tra-1-60, SSEA3, OCT4, SOX2, KLF4 and cMYC. Additionally we perform karyotype analysis, study embryoid body differentiation and analyze germ layer marker expression. To generate the specific neural cell types (oligodendrocytes, astrocytes and neurons) existing protocol have been optimized. The iPS-derived neural cell types are characterized with immunofluorescence and RT-PCR for known markers of the neural cell lineages, and functionally tested in collaboration with others.
The iPS center can assist in the generation of new iPS cells, the differentiation towards and functional analyses of specific neural cell types.
The iPS cell laboratory is an initiative of Prof. dr. M.S. van der Knaap, department of Pediatrics and Child Neurology, VU University Medical Center.
Relevant papers
- Silbereis et al. Olig1 function is required to repress dlx1/2 and interneuron production in Mammalian brain. Neuron, 2014 Feb 5;81(3):574-87.
- Heine VM et al. Tilting the Risk-Benefit Ratio for Preterm Infants. Science Translational Medicine, 2011, Oct 19;3(105)
- Heine VM et al. Small Molecule Smoothened Agonist Prevents Glucocorticoid-induced Neonatal Cerebellar Injury. Science Translational Medicine, 2011
- Heine VM et al. Dexamethasone destabilizes Nmyc to inhibit the growth of hedgehog-associated medulloblastoma. Cancer Research, 2010, 70(13):5220-5.
- Biran V et al. Cerebellar abnormalities following hypoxia alone compared to hypoxic-ischemic forebrain injury in the developing rat brain. Neurobiology of Disease, 2010, 41(1):138-46.
- Maunakea AK et al. Conserved role of intragenic DNA methylation in regulating alternative promoters. Nature, 2010, 466(7303):253-7.
- Heine VM and Rowitch DH. Hedgehog signaling has a protective effect in glucocorticoid-induced mouse neonatal brain injury through an 11betaHSD2-dependent mechanism. Journal of Clinical Investigation, 2009, 119(2):267-77.
- Hyman JM et al. Small-molecule inhibitors reveal multiple strategies for Hedgehog pathway blockade. PNAS, 2009, 106(33):14132-7.
- Schüller U, Heine VM et al. Acquisition of granule neuron precursor identity is a critical determinant of progenitor cell competence to form medulloblastoma. Cancer Cell, 2008, 14(2):123-34.
2026 © iPS Center. All Rights Reserved. Privacy Policy | Terms of Service

